Hi Benjamin. Thanks for your message. This morning I also had the same box pop up with information for with Gen AI webinar instead of the PBBM webinar. However, I did get an email confirmation for the correct, PBBM webinar from Science360<[email protected]>. Please checked your spam folder to see if you indeed received a confirmation email. In the meantime, I am checking in with AAPS and will let you know what I find out. Kind regards, Deanna
------------------------------
Deanna Mudie Ph.D.
Senior Principal Scientist
Simulations Plus, Inc.
Bend OR
[email protected]Disclaimer: Opinions expressed are solely my own and do not express the views or opinions of my employer.
------------------------------
Original Message:
Sent: 02-12-2025 10:40
From: Benjamin Kuzma
Subject: Upcoming webinar - Physiologically Based Biopharmaceutics Model (PBBM) Report Template: Considerations for Improved Quality in View of Regulatory Submissions
Hi All,
I think the registration link might be referencing a previous webinar. I also haven't received a registration / event link for successful registration. Can you confirm the registration link here is for the correct webinar?
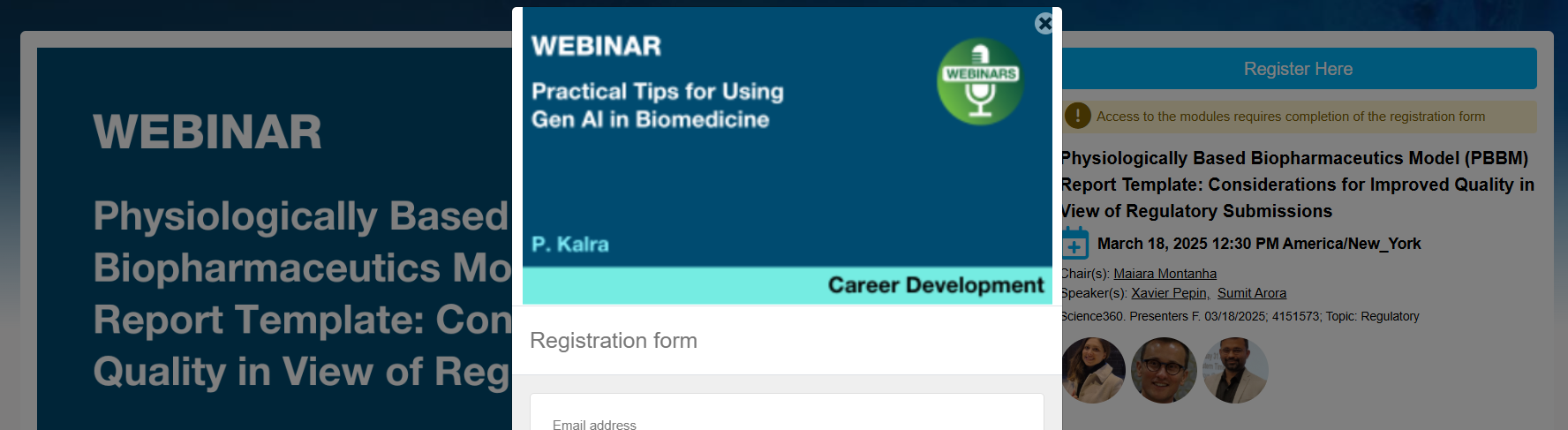
Thanks!
Ben
------------------------------
Benjamin Kuzma
Senior Research Scientist
Vertex Pharmaceuticals Inc
[email protected]
Disclaimer: Opinions expressed are solely my own and do not express the views or opinions of my employer.
Original Message:
Sent: 02-11-2025 15:00
From: Deanna Mudie
Subject: Upcoming webinar - Physiologically Based Biopharmaceutics Model (PBBM) Report Template: Considerations for Improved Quality in View of Regulatory Submissions
I'm excited to announce an upcoming webinar on March 18 organized by the OBAM community, 'Physiologically Based Biopharmaceutics Model (PBBM) Report Template: Considerations for Improved Quality in View of Regulatory Submissions'.
In this webinar Dr. Sumit Arora and Dr. Xavier Pepin will present a summary of current best practices and considerations for PBBM regulatory submissions, with an eye towards decreasing deficiencies, reducing requests for additional information, and improving acceptance rates.
With the growing use of PBBM for guiding drug product development and potentially waiving clinical pharmacokinetic studies, please join us for this impactful topic. Register here: https://science360.aaps.org/science360/2024/webinars-2024/4151573?c=4151573&_zs=0jjtN1&_zl=0i6h7
------------------------------
Deanna Mudie Ph.D.
Senior Principal Scientist
Simulations Plus, Inc.
Bend OR
[email protected]
Disclaimer: Opinions expressed are solely my own and do not express the views or opinions of my employer.
------------------------------