Hi Paul,
There has been a lot of uncertainty on how the IVDR will apply in the context of clinical trials, and I am far from an expert on this topic...
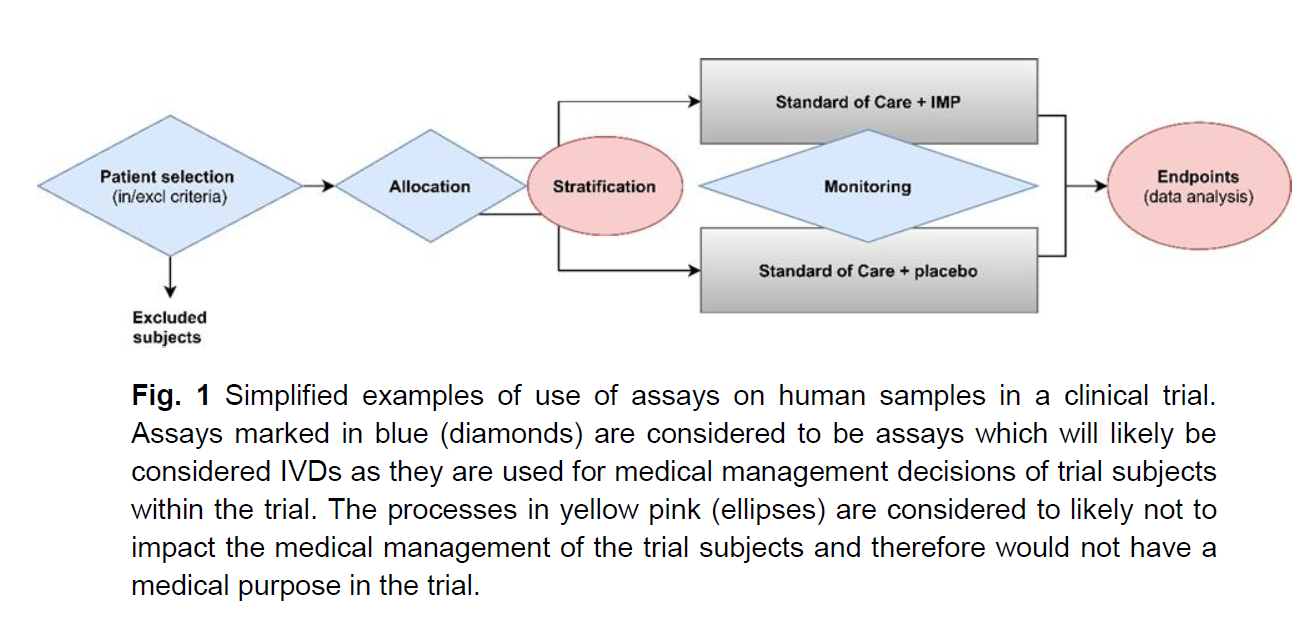
From my understanding, if the assay is used to provide information for medical management decisions, e.g., select patients for enrolment in the trial, assign patients to a treatment arm, guide follow-up measures during and beyond the clinical trial, then it falls within scope of the IVDR.
There's a diagram in the Q&A that seem pretty black and white (or pink and blue...)
------------------------------
Robert Nelson
Associate Director, Scientific Affairs
Labcorp Drug Development
Meyrin Switzerland
[email protected]Disclaimer: Opinions expressed are solely my own and do not express the views or opinions of my employer or other entities to which I am affiliated.
------------------------------
Original Message:
Sent: 06-07-2022 01:26
From: Paul Chamberlain
Subject: Q&A on when the European IVDR regulation apply in clinical trials
Dear Rob,
Thank you for sharing this.
Are you certain that this is applicable to blood sampling for ADA assays used to pre-screen for entry into a trial?
Article 3 states:
3. This Regulation does not apply to:
(b) invasive sampling products or products which are directly applied to
the human body for the purpose of obtaining a specimen;
Thus, I do not see this applicable to blood sampling for ADA assays used to prescreen for entry into a trial.
Could you expand on why you think it is applicable to this particular case?
All the best
Paul
------------------------------
Paul Chamberlain
Immunogenicity Integrated Platform
[email protected]
Original Message:
Sent: 06-03-2022 05:24
From: Robert Nelson
Subject: Q&A on when the European IVDR regulation apply in clinical trials
Hi All,
The attached Q&A was released recently to clarify when the European IVDR regulations (effective 26 May 2022) are applicable to assays used in the clinical trial space. This will likely impact biomarker assays used in precision medicine/patient identification and immunogenicity assays used to prescreen for entry into a trial, amongst many others...
Thanks,
Rob
------------------------------
Robert Nelson
Associate Director, Scientific Affairs
Labcorp Drug Development
Meyrin Switzerland
[email protected]
Disclaimer: Opinions expressed are solely my own and do not express the views or opinions of my employer or other entities to which I am affiliated.
------------------------------